Comprehending the Electromagnetic Spectrum
The electromagnetic spectrum describes the spectrum of electromagnetic waves that range from visible light to the gamma rays. It is a crucial part of science and understanding this area of the universe is crucial. In this article , I am going to discuss some of the most important aspects of this range as well as how they function.
Infrared
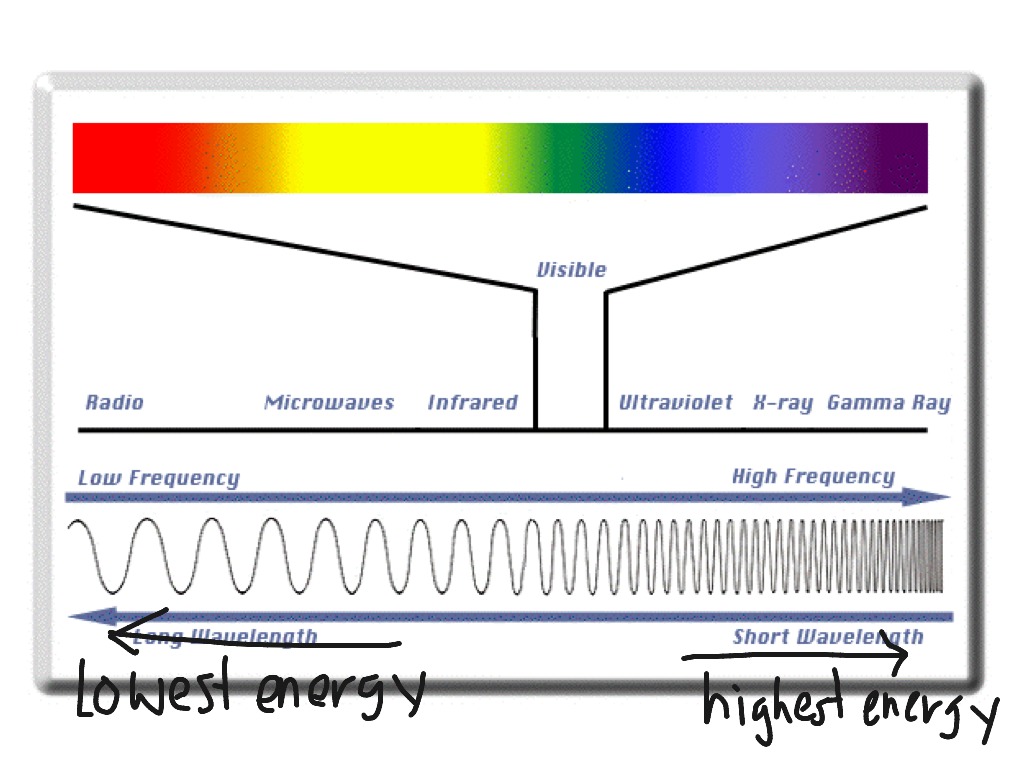
Infrared is an spectrum that radiates electromagnetic radiation which extends beyond the red end of the visible light spectrum. Infrared spectrum is utilized to assess the thermal properties of objects. It is also utilized in night vision equipment.
Generally, infrared is classified into near infrared and far infrared. Near infrared refers to the wavelength that contains the frequencies with the smallest frequencies. These wavelengths are in the range of one to five microns. There are two long and intermediate infrared bands. Each one is distinguished by its own unique wavelengths.
The most famous use of infrared is found in night vision goggles for soldiers. These glasses convert infrared light into visible wavelengths for nighttime vision. Infrared light can also be used for wireless and wired communications.
There isn't any evidence to suggest a connection between infrared and skin cancer. However, it is known that the International Commission on Non-Ionizing Radiation Protection (ICNIRP) has provided guidelines regarding the limit of exposure to invisible visible and infrared radiation.
Visible light
Visible light is a part of the electromagnetic spectrum. The Sun is the main source of light. Some other light sources include the moon and the stars. It is important to know that we cannot see infrared and ultraviolet wavelengths. But, we can see the blue and red light. These colors are mixed in what we call white light.
There are numerous other obscure components of the electromagnetic spectrum, like radio waves and infrared. Some of these have been used for television, radio or mobile phone communications. However, the most effective way to utilize these is to create the right type of filter. This way, we can reduce the harmful impacts of these elements on our body. Additionally, we can build an environment in which we can look at these elements without the use of our eyes.
Although the longest and shortest wavelengths of the visible light could be the most noticeable, the most energy efficient and pleasing to the eye are the shortwave infrared (SWIR) along with microwave.
parts of em spectrum (UV) radiation is a part of the electromagnetic spectrum. It can be used for various purposes. But it can also be harmful. UVB and UVC radiation are not suitable for human eyes, and may cause skin diseases.
This kind of energy is absorbed by molecules and start chemical reactions. The absorbing molecule can then produce visible light, or emit fluorescence.
The spectrum of ultraviolet light is divided into three categories, namely, the extreme, the near, as well as the middle. Common sources for ultraviolet include lasers, arc lamps and light emitting diodes.
Although their wavelengths for UV Rays are smaller that those from X-rays they have more energy. This is useful for breaking bonds in chemical molecules. They are also known in the form of radiation that is nonionizing.
In biochemistry, the ultraviolet spectrum is often used to determine the absorption of a particular substance. There are numerous types of compounds that exhibit significant bands of absorption in the UV.
Ultraviolet light forms a part of the electromagnetic spectrum, and is created by the sun. Its spectrum spans between ten and four hundred nanometres, and its frequencies are between 800 THz and 30 PHz. However, the majority of people do not be able to see it.
X-rays
X-rays are electromagnetic radiation that has high energy. Contrary to gamma and ultraviolet light, X-rays are smaller than visible light and can penetrate thin objects. They are used in a variety different medical procedures, like imaging bone and tissue. There are several kinds of X-rays.

Hard X-rays occur by the collision of an electron with the atom. The result is a void within the electron shell of the atom. Another electron could fill the void. Or, the electron that is incoming could release an atom. In this case, some of the energy from an electron is transferred onto the scattered one.
An X-ray is not to confuse with X-band, which is a low-energy part in the electromagnetic spectrum. Although the two bands are separated by a few centimeters in size, they do not share the same features.
Because X-rays are penetrating and therefore, can be utilized in a myriad of ways. For instance, X-rays can be used in security screening processes to identify cracks in baggage. They are also utilized in radiotherapy for cancer patients. The X-rays can also be used to identify the structural elements of certain materials, such as cement.
Gamma rays
Gamma rays are the most high-energy types in electromagnetic radiation. In actuality, all high energy photons are rays. These photons are produced by nuclear decay and high-energy Physics experiments. They are among the most energetic photons that are found in the electromagnetic spectrum.
Because of their intense energy, gamma radiations are capable of piercing deeply into the materials. The possibility exists for gamma beam to penetrate up to several inches of lead.
A variety of high-energy physics experiments generate gamma rays. For instance a radiation of particles from relativity centered by the magnetic field of the hypernova is visible at 10-billion light years.
Gamma rays can be emitted by the nucleus in some radionuclides following their passage through the process of radioactive decay. Gamma radiation include atomic transitions as well as annihilation and subatomic particle interactions.
Gamma rays in the majority in astronomy come from different mechanisms. Gamma rays from supernovae and nuclear fallout are among the strongest types of electromagnetic radiation. This makes them an excellent source for exploring the universe.
Certain gamma radiations could cause damage to cells within the body. It is good to know that gamma radiations are not as ionizing beta and alpha rays, which means they are less likely to cause cancer. However, gamma rays could alter the DNA's structure and cause burns. Even the smallest amount of gamma rays may cause an ionization of the body.