Chemistry G 9
ASTRUCTURE OF AN ATOM ⚛️
👉Democritus: all matter is composed of tiny INDIVISIBLE particles (he called them atomos...meaning indivisible and hence the name atom .)
👉Aristotle matter is divisible.
📌The idea of these 2 philosophers was not based on experiments.
🔥Here comes JOHN DALTON whose ideas was based in experimental observations.
🔸He proposed Dalton's Atomic theory (back at it later..) using 3 laws of chemical combinations.
🎯Law of conservation of mass : chemical reactions involves only combination and separation of atoms.....
In other words mass of reactants is equal to the mass of products.
🎯Law of definite proportions : chemical compound always contains the same elements in the exact same proportions .for eg.H2O in all three states contains 2 : 1 proportion of H and O by volume respectively .
🎯Law of multiple proportion :when two elements combine with each other to form more than one compound, the weights of one element that combine with a fixed weight of the other are in a ratio of small whole numbers.
🔹Example : by changing the proportion in which N and O combine we can get different compounds (NO,N2O and NO2)
🔥Dalton's is the FATHER OF MODERN ATOMIC THEORY .❗️

🔸Only postulates 2 and 3 are incorrect .
👉Write down the correct version of postulates 2 and 3.✍️✍️✍️
🔴DISCOVERY OF THE SUBATOMIC PARTICLES
1️⃣ ELECTRONS
🔸are the first subatomic particles to be discovered .
🔸Discovered by J.J Thomson using cathode rays discharge tube.
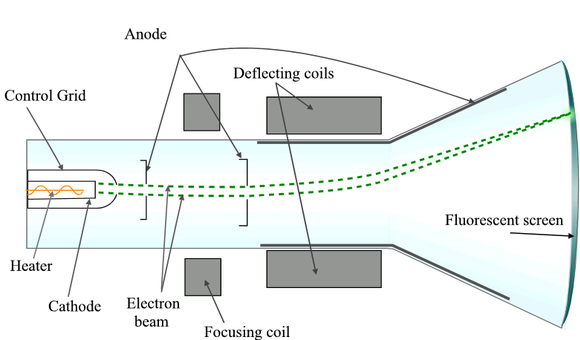
📌cathode rays are stream of electrons leaving the negative electrode (cathode) in a discharge tube.
🔸Those rays were deflected by both magnetic and electric fields .
🔸The cathode rays were deflected to the positive plate in an electric field.(therefore we can conclude that they are negatively charged particles ....opposite charges attract🤗 ....).
🔸Cathode rays travel in a straight line
🔸Thomson used different gases in the discharge tubes however the results were the same therefore electrons are found in all elements . Moreover the electrons in different atoms are essentially the same.😲
🔥electrons retain dual nature of matter they both act like waves( because the diffract and interfere) and particles (because they have mass).
👉Thomson using the mass spectrometer discovered the charge to mass ratio (e- /m) of an electron.
✨e-/m =1.758820 × 10^11 C/kg✨
👉Using the famous oil drop experiment ROBERT MILLIKAN was able to discover the charge of a single electron.
✨Charge of electron = 1.602 x 10^-19 Coulomb.✨
🎯Using the informations we can conclude the mass of a single electron.
✨Mass e = e- / e-/m
=9.11 x 10 ^-31 kg✨
🔥THAT'S ENOUGH FOR TODAY🤗 . we shall continue the discovery of the rest of the subatomic particles in the next part.
🤩https://t.me/euee_preparation..✍️✍️✍️