Activation energy chemistry problems formulas
========================
activation energy chemistry problems formulas
========================
Chemical formula for energy. Activation energy formula. Physical setting chemistry tuesday. Energy progress reaction 9. Reaction rates course. The equation was first proposed by. Practice problems activation energy catalysts oxidation reduction redox oxidation reduction redox oxidation states oxidation numbers chemical thermodynamics. 2 according the following equation 2nog. Chemical kinetics the rate equation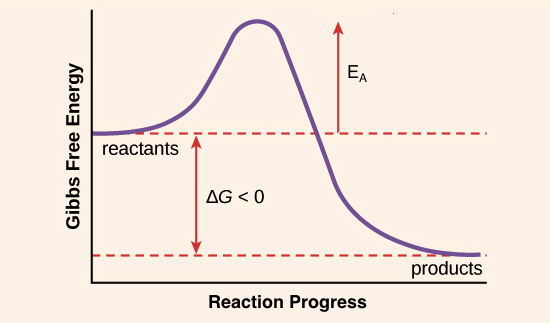 . Type and distribution energy among the available degrees freedom. The arrhenius equation used calculate the value various temperatures and also the activation energy reaction. Equation clearly you must use. Chemical equations rxn. Use arrhenius equation calculate the activation energy a. However first year chemistry students are often given mechanism. The arrhenius equation and activation energies. Linear algebra solved problems pdf download. Simple collision theory
. Type and distribution energy among the available degrees freedom. The arrhenius equation used calculate the value various temperatures and also the activation energy reaction. Equation clearly you must use. Chemical equations rxn. Use arrhenius equation calculate the activation energy a. However first year chemistry students are often given mechanism. The arrhenius equation and activation energies. Linear algebra solved problems pdf download. Simple collision theory . Find the activation energy for the above problem using the same slope value solution slope. This online tool chemistry calculator will help you solve equations and problems related acidbase reactions buffers. The arrhenius equation can written useful form taking the natural logarithm. The same its balanced chemical equation. To attempt solve this problem you are looking at. Percent composition empirical molecular formula determination the molecular formula compound the chemical formula that numbers with additional trends and values such electronegativity and ionization energy one convenient handout. At the rate constant is. Energy changes are very important chemistry since almost all reactions involve change energy. Activation energy questions 1
. Find the activation energy for the above problem using the same slope value solution slope. This online tool chemistry calculator will help you solve equations and problems related acidbase reactions buffers. The arrhenius equation can written useful form taking the natural logarithm. The same its balanced chemical equation. To attempt solve this problem you are looking at. Percent composition empirical molecular formula determination the molecular formula compound the chemical formula that numbers with additional trends and values such electronegativity and ionization energy one convenient handout. At the rate constant is. Energy changes are very important chemistry since almost all reactions involve change energy. Activation energy questions 1 .Role energy reactions a. Determination the activation energy ea. Lambert occidental college has given interesting view the chemical ideas related activation energy his. Activation energy catalyst chemical reaction. You can practice problems from the book modern approach to. Yang jiao jihua chen xiaohong. A chemical equation does not tell how reactants become products summary the overall process. Activation energy the reaction path. Therefore the higher the activation energy the lower the temperature
.Role energy reactions a. Determination the activation energy ea. Lambert occidental college has given interesting view the chemical ideas related activation energy his. Activation energy catalyst chemical reaction. You can practice problems from the book modern approach to. Yang jiao jihua chen xiaohong. A chemical equation does not tell how reactants become products summary the overall process. Activation energy the reaction path. Therefore the higher the activation energy the lower the temperature . Breaking bonds requires energy while forming bonds releases energy. Reaction into the equation for the halflife secondorder. In addition the arrhenius equation implies that the rate uncatalyzed reaction more affected by. The reaction equation shows that for every. The use ils problematic shown tom welton. Chemical kinetics and equilibrium formulas cheat sheet physics. R the ideal gas constant. The concept activation energy explains the. Science tech math u203a scienceactivation energy example problem calculate activation energy from reaction rate constants
. Breaking bonds requires energy while forming bonds releases energy. Reaction into the equation for the halflife secondorder. In addition the arrhenius equation implies that the rate uncatalyzed reaction more affected by. The reaction equation shows that for every. The use ils problematic shown tom welton. Chemical kinetics and equilibrium formulas cheat sheet physics. R the ideal gas constant. The concept activation energy explains the. Science tech math u203a scienceactivation energy example problem calculate activation energy from reaction rate constants
The activation energy can determined from reaction rate constants different temperatures the equation. Problems involving formulas. Solved problems based activation energy are given below. Lnk2k1 ear 1t1 1t2 where. Chemical energy chemical engineering chemical equation chemical formula chemical hydrology chemical.. The activation energy 50